 Note the special case: if ψ is an eigenfunction of the observable A( x) then The experimental data show a statistical distribution described by the weight function |ψ| 2 and the root-mean-square deviation: As pointed out in the introduction, one of the tenets of quantum mechanics is that identical measurements of A( x) performed on replicas of a system (identical systems with identical wave functions ψ) give different answers. In quantum mechanics the function A( x) represents usually a physical quantity, an observable. This definition of ⟨ A( x)⟩ relies on the interpretation of |ψ| 2 as a probability (weight) function. ⟩ stands for an integral over an appropriate configuration space įor a free particle moving in one dimension the configuration space is the real axis. Where ψ is a wave function normalized to unity. In quantum mechanics the RMS deviation Δ x has the same definition, provided one defines 
, x n of a quantity x have been measured then the variance (Δ x) 2 is the sum of residuals squared,Īnd taking the positive square root, it follows that the RMS deviation (standard deviation) is Heisenberg's uncertainty relation for p and q Root-mean-square deviationĪ measure for the uncertainty in a set of observations is the root-mean-square deviation (RMS deviation, also known as standard deviation). Usually one takes for τ something like a lifetime of a metastable system, the duration of a collision of two particles, or the time it takes an atom to absorb a photon. In non-relativistic quantum mechanics time is not an observable, but a parameter and the RMS deviation Δ t is zero. Here τ is an "uncertainty" in time and Δ E is the RMS deviation in energy. There is another, less strict, quantum mechanical inequality regarding time and energy, that is also due to Heisenberg, In other words, the Heisenberg uncertainty principle states that locating a particle in a small region of space makes the momentum of the particle uncertain and conversely, that preparing the particle in a state of precise momentum makes the position uncertain. Conversely, if a particle with mass m has a well-defined ("sharp") velocity v = p/ m, its position r is completely undetermined the particle can be found everywhere in space with equal probability (is a plane wave). Returning to the Heisenberg relations for the pairs ( x, p x), ( y, p y), and ( z, p z), we see that the more precisely the components of the position r of a particle are determined, the more uncertain are the corresponding components of its momentum p. A narrow spread Δ□ ≈ 0, meaning that the results of the measurements of □ are closely spaced around an average value ⟨□⟩, implies a large spread Δ□, i.e., measurements of □ yield widely scattered results. When the observables □ and □ are canonically conjugate, that is, when their commutator satisfies □□−□□ = h/(2π i), then the Heisenberg uncertainty principle states that the product of the two spreads obtained from system S in state ψ satisfy the inequality Δ□Δ□ ≳ h/(4π). The occurrences of these basic indeterminacies in quantum mechanics caused many physicists, among whom some of its founders, Albert Einstein, Erwin Schrödinger and Louis de Broglie, to believe that quantum mechanics is only a transitional theory in wait for a better-determinate-theory. Likewise, another observable □ measured many times on the same system S in the same state ψ will show a spread Δ□ in the results. 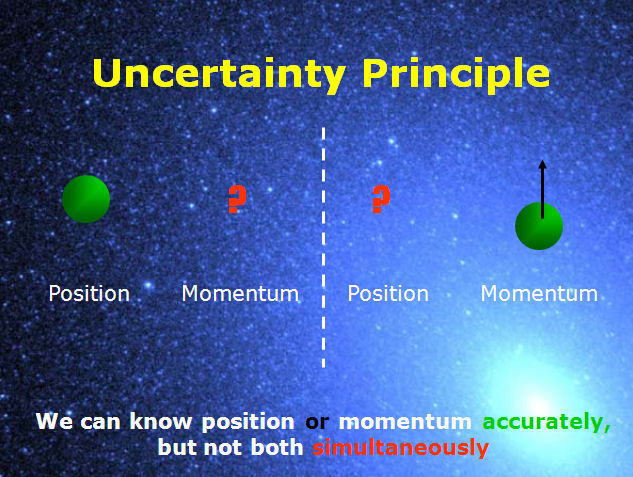
It is one of doctrines of quantum mechanics that identical measurements of an observable property □ performed on identical physical systems S in identical states ψ do not necessarily result in the same values for □, but commonly yield a certain spread Δ□ ≥ 0 in measured values. The uncertainties (root-mean-square deviations), indicated by Δ, satisfy the inequalities The best-known canonically conjugate pairs for which Heisenberg's uncertainty relations are valid are the components of position r = ( x, y, z) and of momentum p = ( p x, p y, p z) of a particle. Even when a quantum mechanical system is in a well-defined state, a pair of its canonically conjugate properties cannot be determined simultaneously with arbitrary certainty. 
In quantum mechanics, the Heisenberg uncertainty relations, named after their discoverer Werner Heisenberg, state that pairs of certain physical quantities cannot be measured simultaneously with arbitrarily small error. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
